Air Quality Monitoring Device

Position
Biomedical Engineer
Year
2023-2024
Company
Northeastern University
This air quality monitoring device was developed as an independent project for use in the College of the Atlantic research involving the renovation of homes in rural Maine and the subsequent changes in air quality. A set of ten devices was developed, each of which contain an Arduino R4 Wifi, an MQ7 CO sensor, and an SEN55 sensor capable of temperature, humidity, PM, NO, and VOC readings to form a device capable of providing accurate and reliable air quality metrics. Additionally, the Arduino Cloud service was used to record and display these values over a Wifi connection for the user, regardless of location.

Intraocular Injection Device
Position
Product Development Engineer
Year
2021-2022
Company
Ximedica | Veranex Solutions
The creation of an intraocular injection device capable of timed drug release to prevent an injection at high velocity potentially traveling through the vitreous humor and damaging the retina. After generating a functioning set of devices, they were used in a formative trial with changes implemented in preparation for clinical trials.
Artificial Womb-Like Device


Position
Product Development Engineer
Year
2021-2022
Company
Ximedica | Veranex Solutions
The creation of an artificial womb-like environment capable of extending the development of the lungs for premature infants once outside of the womb. Additionally, this project involved a priming cart for the initial transfer of a premature infant. A total of three sets of devices were created in preparation for clinical trials.
Tuberculosis LFA Diagnostic Test


Position
Bioengineer, Medical Devices
Year
2022-2023
Company
Northeastern University
Tuberculosis was the leading cause of death due to infectious diseases prior to COVID-19 and the area's most heavily impacted are typically low resource settings without the proper laboratory equipment to effectively test for it. With an average of 10 million tuberculosis cases and 1 million deaths yearly, it was evident that a readily available and affordable test is essential to improving clinical outcomes worldwide. The most used methods of testing are not up to par in low-resource settings which inhibits a physician’s ability to treat patients. Tuberculin skin tests (TST) are the most frequently used test with a sensitivity of 59%, requiring two visits to a clinician and a high rate of false positives. Interferon-gamma release assays (IGRA) are inaccessible in settings with high rates of tuberculosis due to their cost and equipment requirements. Consequently, a novel rapid testing device needed to be developed to provide accurate results at a low cost, to be accessible in low-resource areas without electricity or laboratory equipment, and had to be operable without a clinician.
Laparoscopic Tissue Ablation System


Position
Product Development Engineer
Year
2021-2022
Company
Ximedica | Veranex Solutions
This project revolved around the testing of a laparoscopic tissue ablation device, mainly involving the creation of fixtures compatible with Instron and subsequent changes to the device itself.
Commercial Roofing Drill Mount


Position
Mechanical Design Engineer
Year
2022-2023
Company
Essential Design | PA Consulting
This project involved the modification of a current drill mount for a commercial roofing product that would create a modular mounting system compatible with a variety of commercially available drills/brands.
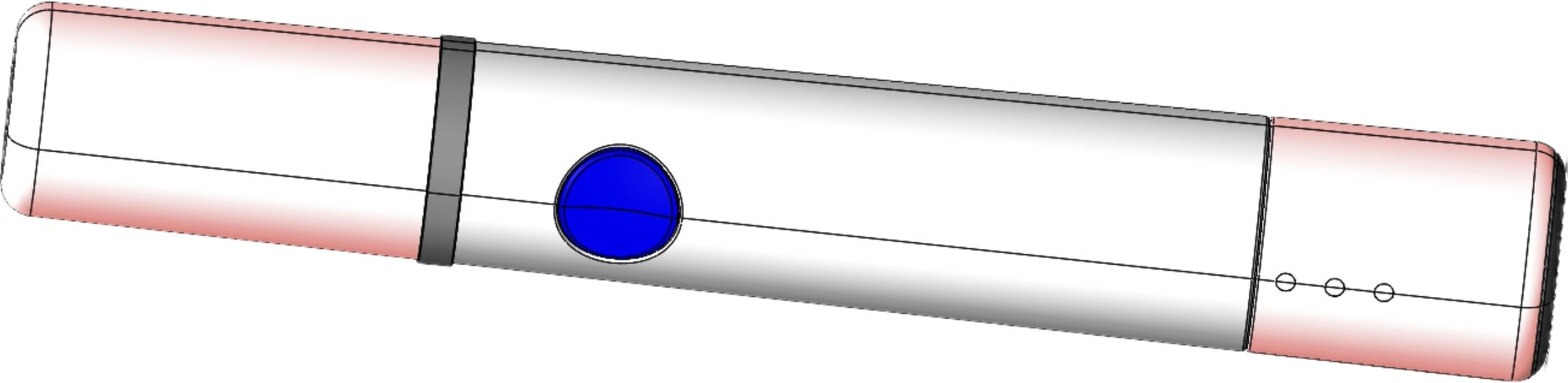
Electro-Current Facial Device
Position
Mechanical Design Engineer
Year
2022-2023
Company
Essential Design | PA Consulting
This electro-current facial device required an update to the housing, creating a tactile yet durable and waterproof button. These updates were made to the device, along with additional changes, to create the next iteration of the Electro-Current Facial device.
Test Fixture - Robotic Surgeon Console

Position
Mechanical Design Engineer
Year
2022-2023
Company
Essential Design | PA Consulting
The creation of this test fixture allowed for further development and testing of the electro-mechanical assembly of a robotic surgeon console. These parts were machined and assembled to facilitate the expedited timeline.